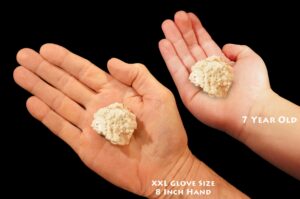
BIOTITE MICA in Andesite 2 1/2″ 2-4 Oz Rough Rock Mineral Specimen Raw Root Chakra Crystal Natural Reiki Stone
$4.99
In stock
Description
This unique stone is flakes of biotite mica embedded in andesite and was mined by a friend of mine from Union Valley Reservoir, El Dorado County, California. The biotite mica sparkles as you turn the stone in the light. The golden mica and light gray andesite make beautiful contrasting colored specimens.
NOTE: Tiny flakes will come off in shipment no matter how well it is packaged. That is just the nature of biotite and andesite.
You will receive one specimen from the lot shown in the photos that is 2 inch to 2 1/2 inch and weighs 2 to 4 ounces.
Bulk Purchase Listing
You are buying one rock from the lot shown in the photographs. Listing every rock separately adds a lot in labor costs. We pass that savings on to you with lower prices and higher quality than the competition. 75% of all of our Reviews are from customers amazed by what they received buying our bulk purchase listing products!
Kidz Rocks – The Best Value on the Internet!
Higher quality, larger size, lower price genuine natural rocks and minerals with customer service second to none. Buy ANY 5 or more items and get 30% off your entire order and a FREE surprise specimen! Maximum $8.75 shipping no matter how many items are on your order and no return required refunds!
Mineral Description
Biotite is a common rock forming mineral, being present in at least some percentage in most igneous and both regional and contact metamorphic rocks. Biotite mica is sometimes called iron mica because it is iron-rich. It is also sometimes called black mica as opposed to white mica (muscovite) both form in some rocks, in some instances side-by-side.
Sheets of biotite are flexible they can be bent and they go back to their original shape. Mica possesses great resistance to the passage of electricity and heat and is therefore used as an electronic insulator.
Andesite is volcanic rock named after the Andes Mountains and is the name of a family of fine-grained, extrusive igneous rocks that are usually light to dark gray in color. They have a mineral composition that is intermediate between granite and basalt.
About Kidz Rocks
Since 2009, Kidz Rocks, a USA company, is “The Best Value on the Internet” providing higher quality, larger size and lower price rocks, minerals and crystals with customer service second to none.
Kidz Rocks Promise:
We are committed to redefine what it means to shop for rocks and minerals online. NO insane high prices, NO great piece one time and then a disappointing piece next time, NO rocks that look big online and then a tiny piece shows up, NO waiting a long time for shipping …
A partnership that you can trust and count on with each and every order that you will be pleased if not amazed when you open your package! Always high quality – Always larger sizes at lower prices that other dealers ƒ?? Always a positive and fun shopping experience.
If it is not a specimen we would add to our own personal collection, we don’t ship it out to you! When you buy from us you will be satisfied with each and every purchase or your money back and you keep the rocks!
Please feel free to call us at 1-818-518-6278 10 AM to 10 PM 7 days a week for wholesale discounts and special requests, we can get pretty much any rock on the planet. We ship all orders USPS, weekday orders ship the same day they are placed, weekend orders ship on Monday’s. We disclose any treatments done to the rocks after they are mined in our listing descriptions. If no treatments are stated then it is exactly as it came out of the ground.
Thanks,
Alan & Jaidon
Additional information
| Weight | 0.25 lbs |
|---|
1 review for BIOTITE MICA in Andesite 2 1/2″ 2-4 Oz Rough Rock Mineral Specimen Raw Root Chakra Crystal Natural Reiki Stone
Only logged in customers who have purchased this product may leave a review.








Vivian Johnson (verified owner) –
Beautiful! Exactly as described, well packaged, fast shipping!! Thank you KidzRocks!!!